
Some of the groups have widely used common names, including the alkali metals (group 1. If the Group 1 elements all have one s s electron in their outer orbital, we can. The name alkaline comes from the fact that compounds of these elements form basic (pH greater than 7) or alkaline solutions when dissolved in water. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 118 from left to right) the seven horizontal rows are called periods. Group 2 elements are referred to as alkaline earth metals (tan column below). Text and images Copyright (c) 2010 by Theodore W. The periodic table is an arrangement of the elements in order of increasing atomic number. The others are soft metals that react violently with water to release hydrogen gas, which either bubbles off or spontaneously ignites depending on how far down you are on the table.Ĭlick any element below to see all the samples of that element. Hydrogen is an anomaly: It's traditional to put it in this column, but it really has very little in common with the rest. As a rule, you wouldn't want any around the house in pure form, though as a compound they can be quite tasty. Even after the periodic nature of elements and the table itself were widely accepted, gaps remained. 2: Elements in the periodic table are organized according to their properties. The exception is hydrogen, which behaves as a nonmetal at room temperature and pressure and is found on the upper left corner of the periodic table. The Alkali Metals comprise the left-most column of the periodic table. For the table to fit on a single page, parts of two of the rows, a total of 14 columns, are usually written below the main body of the table. The nonmetals are in the minority on the periodic table, mostly located on the right-hand side of the periodic table. The rows are arranged so that metals are on the left side of the table and nonmetals are on the right side. 
The seven rows of the periodic table are called periods. Nonmetals are separated from metals by a line that cuts diagonally through the region of the periodic table containing elements with partially filled p orbitals.The collection Alkali Metals in the Periodic Table H The periodic table is a tabular arrangement of chemical elements that is arranged by increasing atomic number and groups elements according to recurring properties. Other categories are possible, depending on the criteria for inclusion. Alkali metals are any of the elements found in Group IA of the periodic table (the first column). The nonmetals are located on the upper right side of the periodic table. The Metallic elements table in this section categorises the elemental metals on the basis of their chemical properties into alkali and alkaline earth metals transition and post-transition metals and lanthanides and actinides. Nonmetals include solids, liquids, and gases at room temperature and pressure. Alkali metals are s block elements that are located on the left-hand side of the periodic table.They tend not to be malleable or ductile, so they form brittle solids. Nonmetals are typically poor conductors of heat and electricity, with low melting and boiling points.For example, diamond and graphite are both pure carbon. Most nonmetals take many forms called allotropes.Hydrogen is unique in that it is generally placed in group 1, but it is not a metal. Nonmetals typically don't look metallic. The alkali metals are lithium, sodium, potassium, rubidium, cesium, and francium. 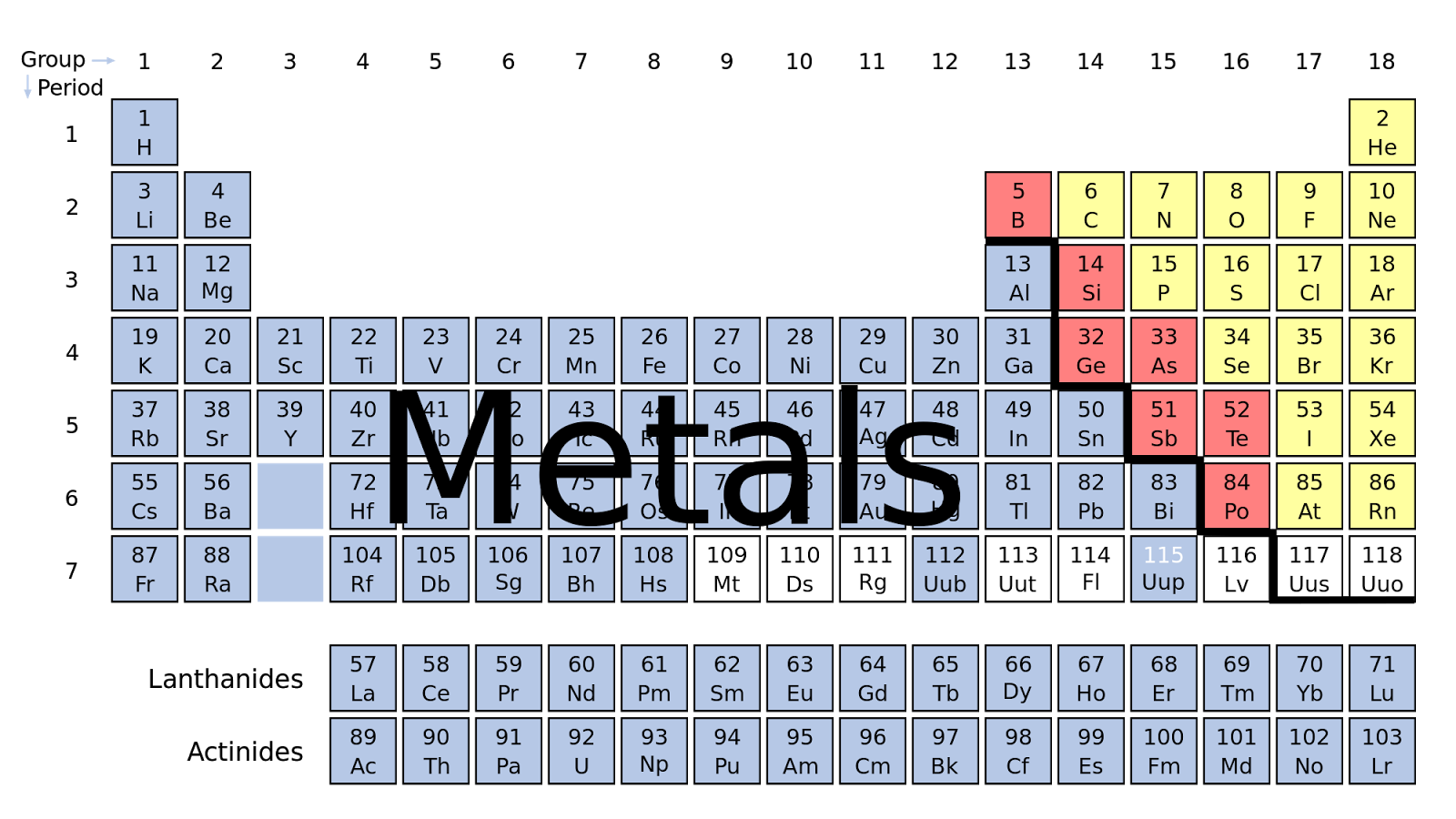
The nonmetals include the group called the nonmetals, as well as the halogens and the noble gases.All of the alkaline earth metals tend to form 2+ cations (pronounced KAT-ieunz). CATION: The positive ion that results when an atom loses one or more electrons. The alkali metals all have one valence electron in thes1 orbital, and are highly reactive. Group one elements share common characteristics. Group 1 of the periodic table of elements consists of hydrogen, and below it the six alkali metals: lithium, sodium, potassium, rubidium, cesium, and francium. This family consists of the elements lithium, sodium, potassium, rubidium, cesium, and francium (Li, Na, K, Rb, Cs, and Fr, respectively). Alkali metals are powerful reductants and so do not exist as the free metal in the relatively oxidizing environment at the earths surface. These elements are located on the righthand side of the table, with the exception of hydrogen. The elements in Group 1 of the periodic table of elements, with the exception of hydrogen. The alkali metals make up Group 1 of the periodic table.The nonmetals are a small group of elements on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
